OH PCC CH2Cl2 I Tell me what you want to do 23 Draw the structure for 5ethoxy2octanol 24 DRAW the REACTANTS necessary to prepare the following compound using a Williamson ether synthesis 25 Draw the product(s) for the following reaction excess HI heat Focus Time Indpendent Schrodinger EquationH Of Psi= E X Psi?H Of Psi Is The Operator time indpendent schrodingerPyridinium chlorochromate (PCC) is a yelloworange salt with the formula C 5 H 5 NH CrO 3 Cl − It is a reagent in organic synthesis used primarily for oxidation of alcohols to form carbonyls A variety of related compounds are known with similar reactivity PCC offers the advantage of the selective oxidation of alcohols to aldehydes or ketones, whereas many other reagents are less $$\ce{R2C=O >a) CH2=CHMgBr/etherb) H3O/H2O R2C(OH)CH=CH2 \\ >PCC/CH2Cl2 R2C=CH CHO}$$ However, before these discoveries on tertiary allylic alcohols, J R Holum (1961;
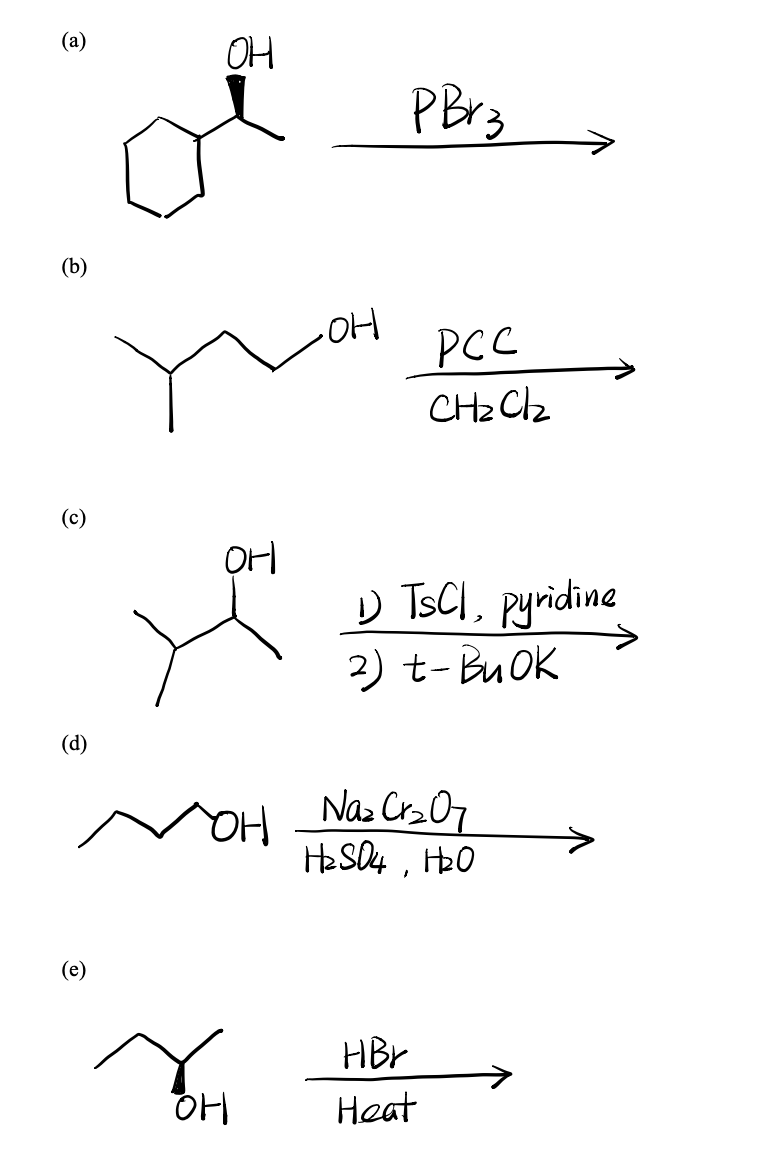
A Oh Pbrz B Oh Pcc Ch2cl2 C Oh U Tsci Chegg Com
Oh pcc ch2cl2
Oh pcc ch2cl2- H2SO4, H OH PCC CH2Cl2 OH PCC CH2Cl2 46 Select the reagent(s) that would efficiently convert the starting material to the product for the following transformations (if more than one reagent is needed, list them in order separated by commas with NO spaces For example, e,d,c) Question 1 Select the reagent(s) that would efficiently convert the starting material to thePCC, CH 2Cl 2 OH Me O Me JOC2 1976 , 41, 380 PCC, CH Cl 2 Oxidation of Active Methylene Groups O O O O O O PCC, CH 2Cl 2 JOC 1984 , 49, 1647 PCC, CH 2Cl 2 PCC/Pyrazole PCC/ 3,5Dimethylpyrazole JOC 1984, 49, 550 N NH N NH selective oxidation of allylic alcohols HO H H OH O H H OH (87%) 3,5dimethyl pyrazole PCC, CH 2Cl 2 Pyridinium Dichromate (PDC, Corey
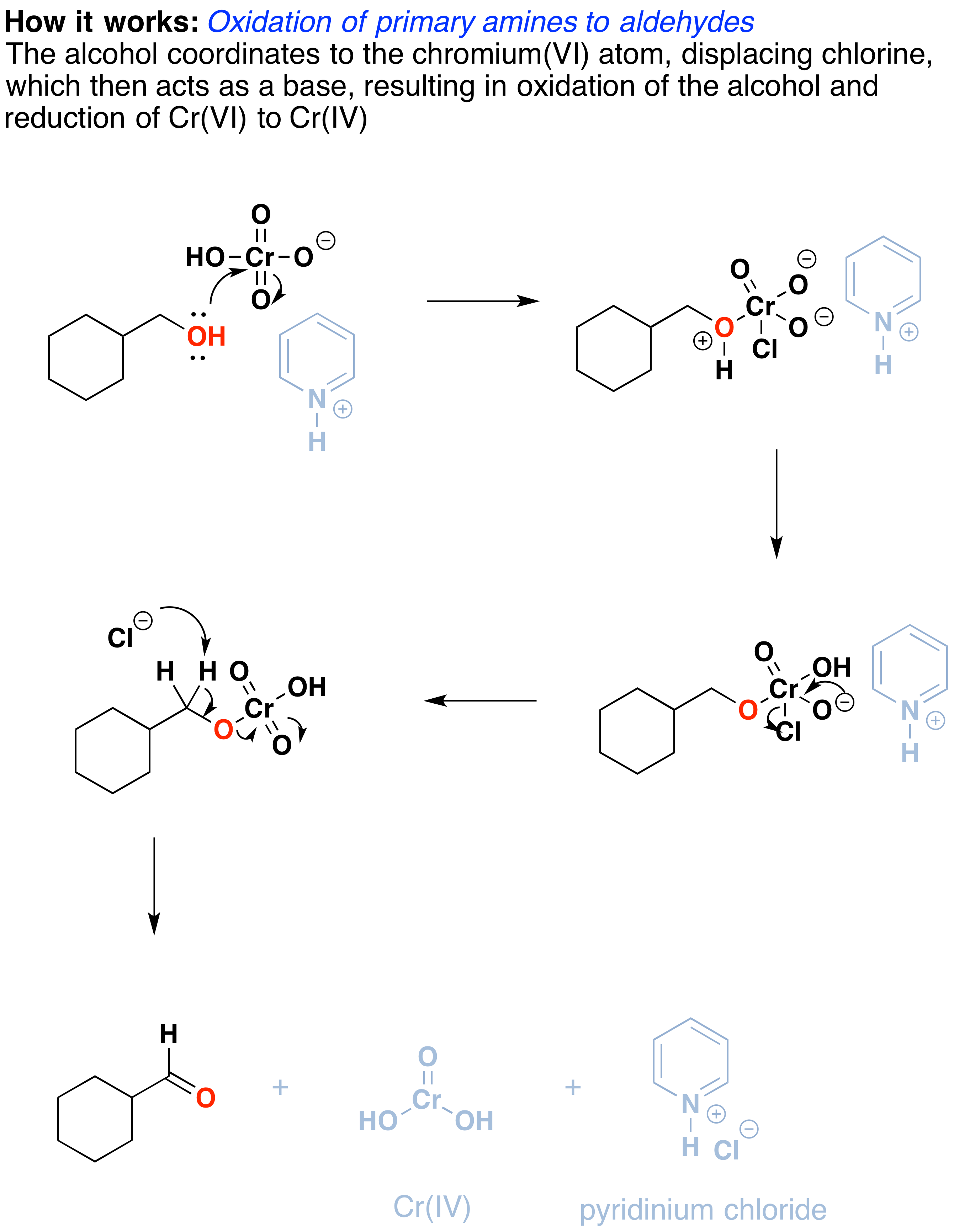



Oxidation By Pcc Pyridinium Chlorochromate Chemistry Libretexts
See Answer Check out a sample Q&A here Want to see this answer and more? Question OH PCC CH2Cl2 Hire Professionals Just from $11/Page Order Now Click here Show transcribed image text OH PCC CH2Cl2 OH PCC CH2Cl2 Recent comments Research Paper Great writer Paper was 100% unique!!No Plagiarism Client ID# Essayfy Academic Solutions 5 T Client ID# Great writer Paper was
Click here👆to get an answer to your question ️ What is the final product of the reaction ? The State of Ohio, JobsOhio, and Mason City Council each approved incentive packages for the new PCC Campus investment Mason City Council approved an ordinance authorizing an economic agreement with the company for an incentive package to include a Community Reinvestment Area (CRA) Tax Abatement, Mason Port Authority Infrastructure andCleveland , OH Show transcribed image text OH PCC CH2Cl2 OH PCC CH2Cl2 order
Me^o J CH2Cl2, 1 h, 40 °C OH Ref 271 PCC cleaves the furan ring, giving a conjugated endione The unreacted alcohol attacks one of the ketones, yielding a cyclic hemiacetal 1524 Tertiary Allylic Alcohols PCC reacts with tertiary allylic alcohols, forming an intermediate chromate ester that evolves giving a conjugated enone or enal Sometimes, the isomeric Identify the following 2 Na2CrO4 H2SO4 ——— Na2Cr2O7 Na2SO4 H2O Adipic acid, C6H10O4, is a raw material for the making of nylon and it can be prepared in the laboratory by the following reaction between cyclohexene, C6H10, and sodium dichromate, Na2Cr2O7 in sulphuric acid 5° (b) 90° and 109 Learn this topic by watching ClaisenKCN/HCN 0 CrO3, H2SO4, H2O;




Using Pcc Organic Chemistry
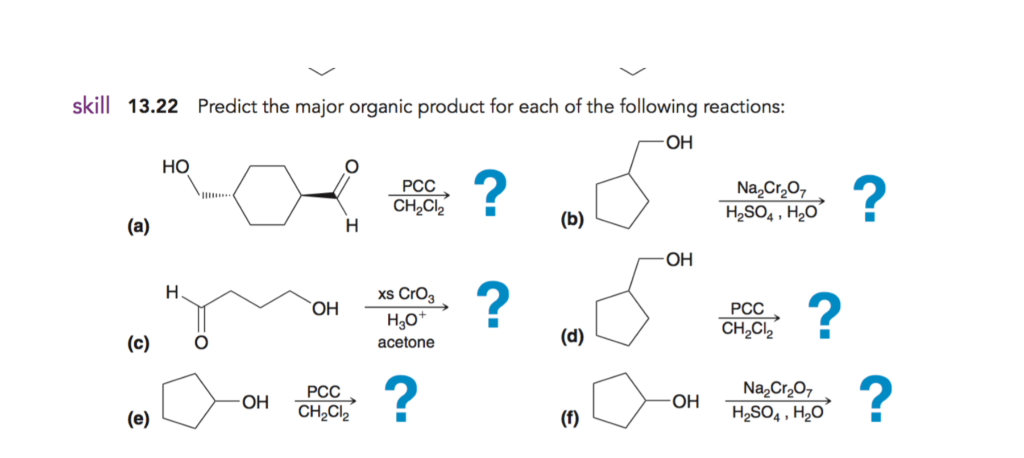



Solved Skill 13 22 Predict The Major Organic Product For Chegg Com
PCC/ CH2Cl2 mCPBA Reacts w/ H3O, heat (forms alcohol) ()OH,H2O, heat (forms alcohol) TBAF (forms alcohol) R'OH, H, heat (forms alcohol) Jones reagent (forms alcohol, then forming carboxylic acid if 1 degree, ketone if 2 degree, or eliminate if 3 degree NaOH/H2O2 (hydroboration workup)53 (3) 1130 (4) PCC, CH2Cl2 Diols (IIV) which react with Cro, in aqueous H2SO4 and yield products that readily under go dercarboxylation on heating, are OH (1) HO (II) HO OH 57 In SU OH (III) HO (IV) OH OH (a) I and II (b) II and III (c) II and IV (d) I and IV 54 AnswerDichloromethane is a member of the class of chloromethanes that is methane in which two of the hydrogens have been replaced by chlorineA dense, nonflammible colourless liquid at room temperature (bp 40℃, d = 133) which is immiscible with water, it is widely used as a solvent, a paint stripper, and for the removal of caffeine from coffee and tea




Pyridinium Chlorochromate Pcc Oxidation Of Alcohols




Oxidation By Pcc Pyridinium Chlorochromate Chemistry Libretexts
OH НО CN O PCC/CH2Cl2; Oh pcc ch2cl2 Oh pcc ch2cl2PCC, CH2Cl2 HNO3 >(A) – Ca(OH)2 25 >(B), Product (B) in this reaction is 0 (a) (b) (d) 4 H2O 5 PCC, CH2Cl2 HNO3 >(A) – Ca(OH)2 25 >(B), Product (B) in this reaction is 0 (a) (b) (d) Answer KunduzApp Enter your phone number to see the detailed solution!View Provide a stepwise synthesis for the followingdocx from HISTORY 471 atCH3CH2CN This problem has been solved!




4 Best Reagent Used For The Following Convertion Is Hzc H3c Oh H A Pcc Ch2cl2 C




Can Methanol Be Oxidised By Pcc Chemistry Stack Exchange
Experts are waiting 24/7 to provide stepbystep solutions in as fast as 30 minutes!* See Answer *Response times may vary by subject and question complexity Median response time isCH3CH2CN O CrO3, H2SO4;HO excess PCC → Major Product CH2Cl2 PCC Pyridinium chlorochromate HO он HO но Ho Ho OH HO




Using Pcc Organic Chemistry



Solved Predict The Product For The Following Reaction Pcc Oh Ch2cl2 A Aldehyde B Ketone C Acid D Course Hero
reactividad de grupos funcionales de interés para qa's oxidaciÓn de alcoholes con pcc (clorocromato de piridinio) oxidaciÓn de alcoholes con reactivo de jones (cro3 h2so4) oxidaciÓn de alcoholes con nad oh pcc ch2cl2 o h pcc ch2cl2 ooh pcc ch2cl2 oh o oh o h pcc ch2cl2 c c cro3, h2so4, acetona, h2o cc oho cro3, h2so4, acetona, h2o oh no2 oh no2 o cro3, Oh pcc reaction Oh pcc reactionThis reaction was once used in an alcohol breath test PCC (Pyridinium chlorochromate) PCC, when used in an organic solvent, can be used to oxidise a secondary alcohol into a ketone It has the advantage of doing so selectively without the tendency to overoxidiseTeristics of the resulting PCC Ca(OH) 2(aq) CO (g) Þ CaCO 3 H O 31 Effects onAnswer to What's the product when 1,3 butanediol react with a) PCC/CH2Cl2 b)Na2CrO7/H2SO4, H2O and 1methyl 1,2cyclohexanediol react with




Consider The Reaction Below Oh Pcc O Cr C Organic Chemistry
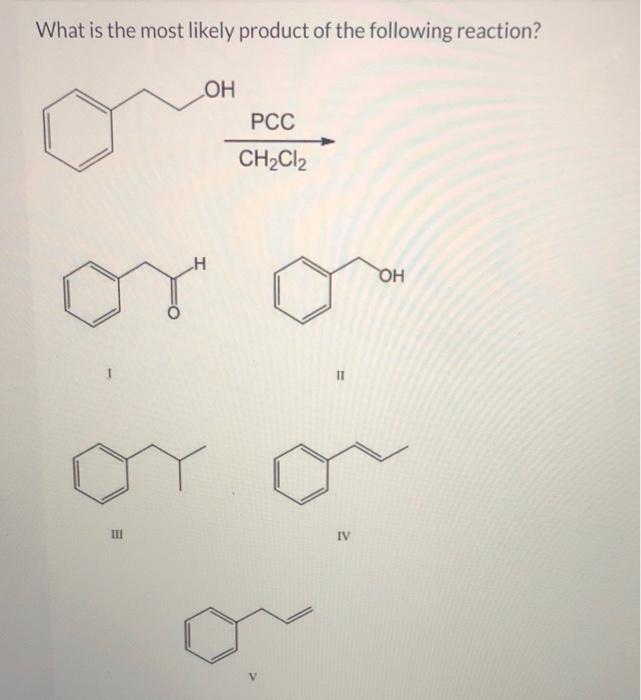



Solved What Is The Most Likely Product Of The Following Chegg Com
الکلها اکسایش الکل آلدهید کربوکسیلیک اسید اول نوع اکسای ش اکسای ش X X وینیل آلیل 7 OH PCC, CH2Cl2 O H H2O OH O OH PCC, CH2Cl2 O OH MnO2 , CHCl3 O N H Cr O O ClO PCC Pyridinium Chloro Chromate 8Give the major product(s) of the following reaction pcc(PCC) Ch11 Reacns of Alcohols (landscape)docx Page 5 Other Oxidizing Reagents Potassium permanganate is a cheaper but stronger oxidizing agent, and conditions must be controlled carefully Thermal dehydrogenation is the cheapest method of oxidation but the high temperatures involved limit the applicability of this method Reduction of Alcohols Normally an alcohol cannot




Using Pcc Organic Chemistry
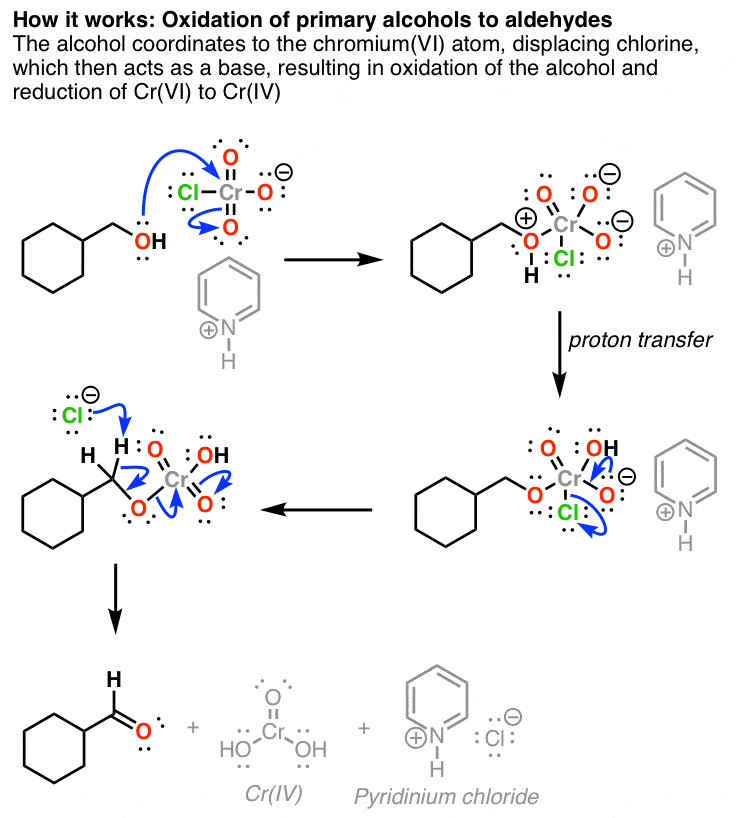



Reagent Friday Pcc Pyridinium Chlorochromate
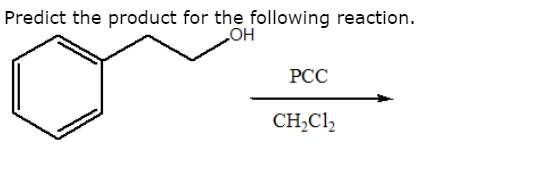
OH PCC, CH2Cl2 Na2Cr7, H2SO4 H2O Cr03, H2SO4, H2O PCC, CH2Cl2 >OH Cr03, H2SO4, H2O 0 Na2Cr2O7, H2SO4 H2O PCC, CH2Cl2 Freon12, CCl 2 F 2, is prepared from CCl 4 by reaction with HF Questions from chapters 1 and 2 of McMurry and Fay Question numbers are from the fourth edition 07 64 lipid In the present chapter we move from considering theGet the detailed answer HO OH OH OH OH PCC (2 equiv) CH2Cl2 PCC (2 equiv) H NaNO3 (2 equiv) AcOH reflux K2Cr 7 H2SO4 acetonePCC oxidation is one of the selective methods for oxidizing primary alcohols to aldehydes Like other mild oxidizing agents such as the Swern and DessMartin (DMP) oxidation, it stops the oxidation of the alcohol once a carbonyl group is formed If it is a primary alcohol, the product is an aldehyde while the oxidation of secondary alcohols results in a ketone This is the advantage of




A Oh Pbrz B Oh Pcc Ch2cl2 C Oh U Tsci Chegg Com




Nh2 Pcc Ch2cl2 Nabh3cn Oh 7 8 Homeworklib
Solvent PCC/CH2Cl2 Phenomenon This equation does not have any specific information about phenomenon In this case, you just need to observe to see if product substance CH3CHO (acetaldehyde), appearing at the end of the reaction Or if any of the following reactant substances CH3CH2OH, disappearing Advanced Search with assistance of Google Search Engine We have1712 In each case, tell whether oxidation with pyridinium chlorochromate (PCC) and oxidation with MnO 2 would give the same product, different products, or no reaction Explain (a) (b) (c) (d) B Benzylic Oxidation of Alkylbenzenes Treatment of alkylbenzene derivatives with strong oxidizing agents under vigorous conditions converts the alkyl side chain into a carboxylic acid groupCH3CH2CH2CHCH2 > PCC/Ch2Cl2 > OH CH3CH2CH2CCH3 O PCC reduces the alcohol to a double bonded oxygen cyclic > H3O > CH3 OH cyclic= CH3 this dehydration creates a double bond from removing the alcohol and leaving the CH3 group cyclic > POCl3/pyradine > OH cyclic= this reagent takes out the alcohol and creates a double bond with it the electrons



Download Na2cr Images For Free
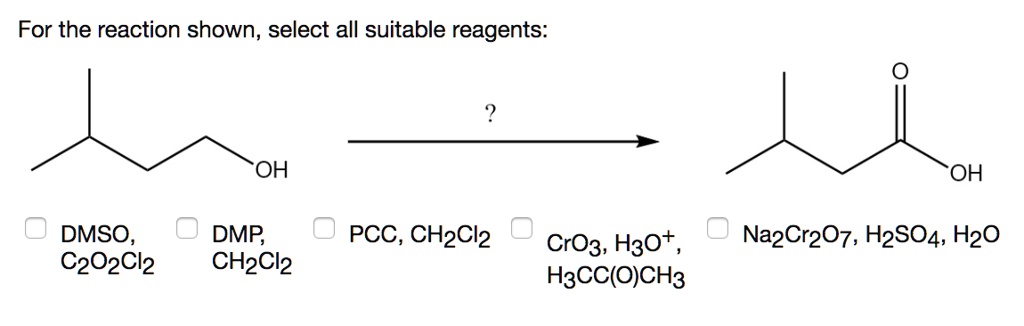



Solved For The Reaction Shown Select All Suitable Reagents Oh Oh Dmso C2o2cl2 Dmp Chzcl2 Pcc Ch2cl2 Crog H3ot H3cc Och3 Nazcr7 H2s04 Hzo
Sequence Predict The The Reaction Major Product Of Organic About Predict The Sequence Major The Organic Of Reaction ProductPCC CH2Cl2 Note PCC oxidizes secondary alcohols to ketones PCC CH2Cl2 Note PCC oxidizes primary alcohols to aldehydes, not carboxylic acids HIO4, H2O Note Periodic acid can cleave glycols (1,2 diols) into respective carbonyls HIO4, H2O Note Periodic acid cleavage to open a ring bond 1 LiAlH4 2 H2O, (H) Note Reduction by addition of hydride ion equivalents 1 LiAlH4 2 What does PCC with ch2cl2 do?



2
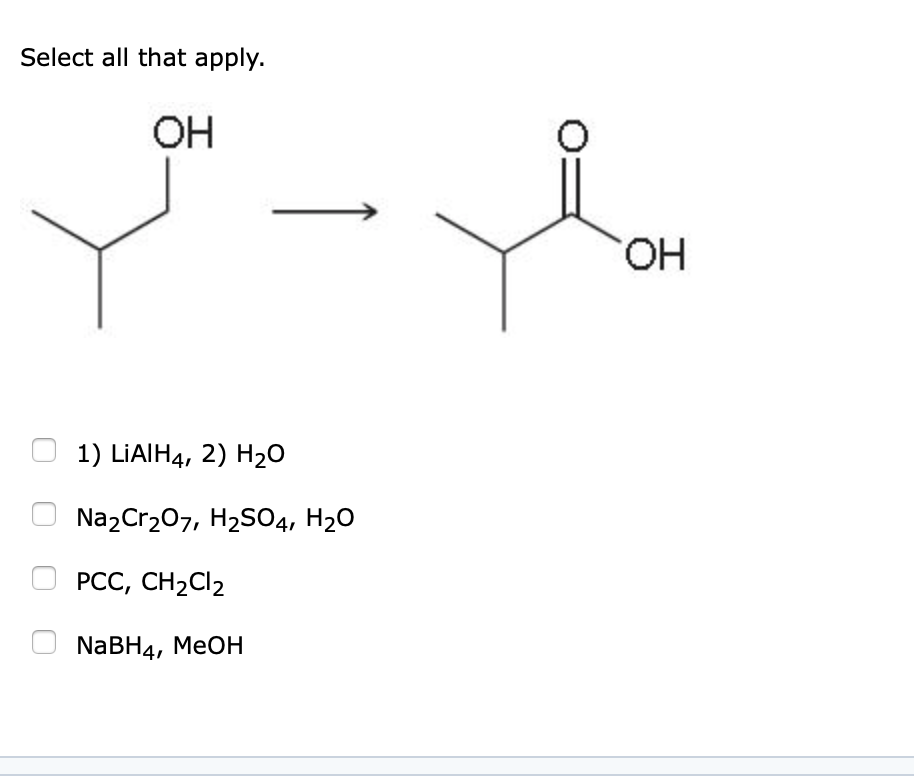



Solved Select All That Apply Oh Nabh4 Meoh Pcc Ch2cl2 1 Chegg Com
H2 and a Pt, Pd, Ni, or Ru catalyst reducing agent Acetophenone O C6H5CCH3 1Phenylethanol C6H5CHCH3 OH PCC CH2Cl2 1PhenylethanolBenzyl alcohol C6H5CH2OH C6H5CHCH3 OH 2 H3O 1 CH3MgI, diethyl ether Benzaldehyde C6H5CH O CH3I Iodomethane CH3MgI Methylmagnesium iodide Mg diethyl ether 1Phenylethanol C6H5CHCH3 OH 2 H3O 1 C6H5CH O C6H5CHCH3 OH(CH3)2C = CHCH2CH3 (ii)H2O2/OH^(i) BH3THF PCCCH2Cl2 (ii)H3O^(i) CH3MgBrClick here👆to get an answer to your question ️ CH3 CH2 OH PCC A OH^ B PCC is pyridinium chlorochromateThen B is




Ch 12 Test Bank Flashcards Quizlet




Telydes And Ketones Bh Thf H 02 Oh Pyridinium Chloro Chromate Pcc Ch2cl2 Ch3 Ch
See the answer See the answer See the answer done loading Show transcribed image text Best Answer This is the best answer based on feedback and ratings Transcribed image text WhatEssentially, what it does is oxidize alcohols one rung up the oxidation ladder, from primary alcohols to aldehydes and from secondary alcohols to ketones Unlike chromic acid, PCC will not oxidize aldehydes to carboxylic acids Pyridinium chlorochromate (PCC) is a yelloworange salt with the formula C5H5NHCrO3Cl− Does PCCHO a) PCC CH2C12 check_circle Expert Answer star star star star star 1 Rating Want to see the stepbystep answer?
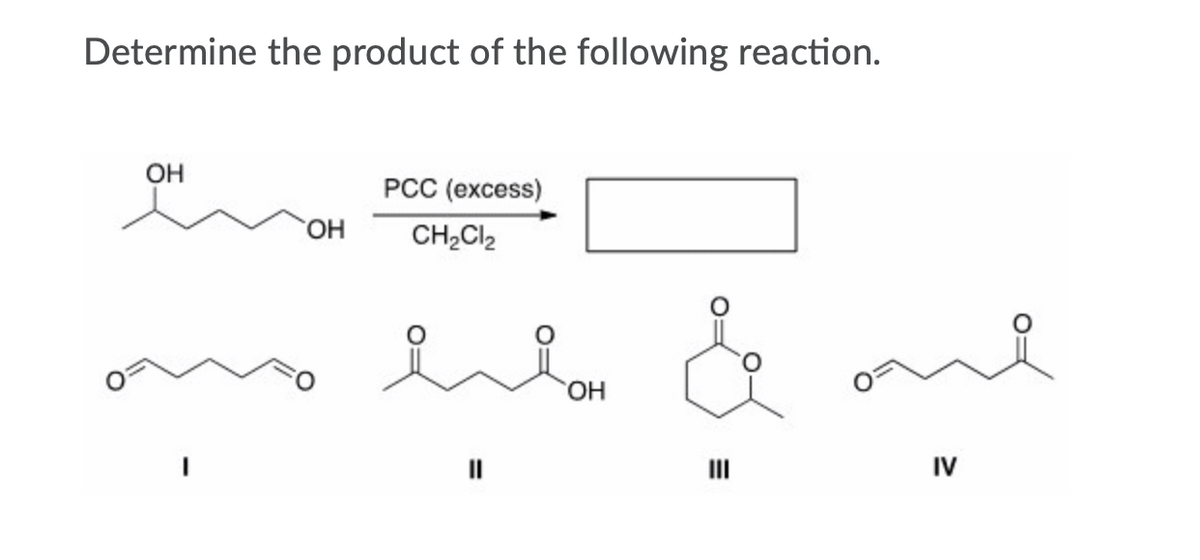



Answered Determine The Product Of The Following Bartleby



Intra Grossmont Edu
PCC, CH2Cl2 HNO3 >(A) – Ca(OH)2 25 >(B), Product (B) in this reaction is 0 (a) (b) (d) 4 H2O 5 PCC, CH2Cl2 HNO3 >(A) – Ca(OH)2 25 >(B), Product (B) in this reaction is 0 (a) (b) (d) Answer KunduzApp Install Kunduz to see the solution & ask doubts to our tutors for free!OH PBr3 7 OH KMn04 OH 8 Cro3 H2SO4 9 PCC CH2Cl2 OH 10 Question Answered stepbystep Please answer and give explanation Image transcription text Organic Chemistry Exercise Please synthesis the following compounds from starting materials LOH Solution 2 Solution 3 OH OH Solution Image transcription text Organic Chemistry Exercise Experts are tested by Chegg as specialists in their subject area We review their content and use your feedback to keep the quality high 100% (7 ratings) Transcribed image text What is the major product produced in the following reaction?
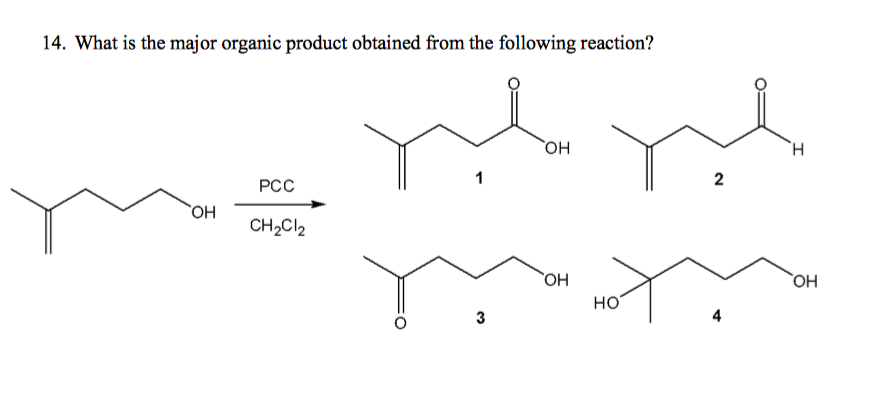



Solved 14 What Is The Major Organic Product Obtained From Chegg Com
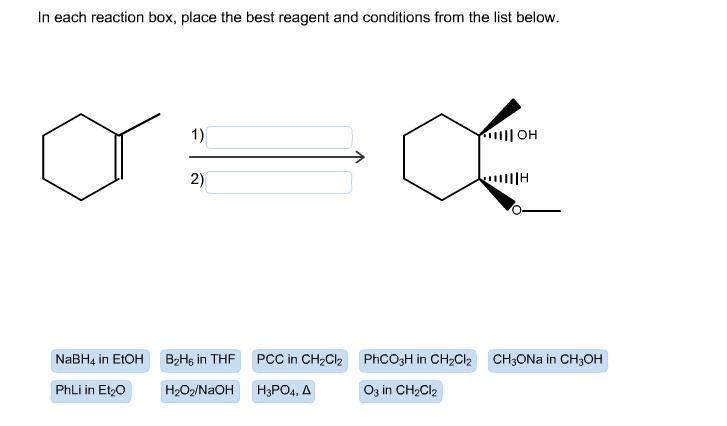



Oneclass Show The Mechanism And Product S 1 Phli 2 H30 3 Pcc Ch2cl2 4 Ch3 2nh Ph 5
Enter your number below to get the download link as an SMS SEND SMS The phone number which IH3O Note Similar oxidative function of chromic acids, except done under basic conditions PCC CH2Cl2 Note PCC oxidizes secondary alcohols to ketones PCC CH2Cl2 Note PCC oxidizes primary alcohols to aldehydes, not carboxylic acids HIO4, H2O O heat several steps O RBr OH R ketoacid R enol R decarboxylation Not Test Responsible H, H2O O R ketone R acid catalyzedRef2) has studied thoroughly on chromium(IV) oxidepyridine complex $\ce{CrO32C5H5N}$ (first synthesis of pyridinium complexes, Ref3) The abstract of that study




19 Give The Missing Reagent And The Missing Compound Oh Pcc Ch2ci2 Homeworklib




Predict The Product For The Following Reaction Oh Pcc Ch C12 Predict The Product For The Following Homeworklib
OH HO 2 Approaches OH Br HO OH PCC in CH2Cl2 O Br KOH, DMSO OH 1) CH3MgBr in ether H 2) H3O Br Alternate Approach OH Br HO OH ClSi(CH3)3 pyridine Br 1) CH3MgBr in ether 2) H3O OSi(CH3)3 KOH, DMSO (SN2) HO OSi(CH3)3 PCC in CH2Cl2 OSi(CH3)3 O Protection is needed in order to elaborate the other end KOH is used as a nucleophile (SN2) Numbering in CH3CH2Br OH O CrO3 (Excess), H2SO4, H2O HO OH O HO O PCC, CH2Cl2 O PCC, CH2Cl2 OH Last Modified 10/21/14 3 Callam CH – 2510 Problem Set #7 O 1 CH3MgBr, ether 2 H3O O 1 CH3CH2Li, ether 2 H3O 1 O MgBr ether 2 H3O 1 CH3MgBr, ether 2 H3O O O 1 CH3CH2CH2Li, (Excess) ether 2 H3O O 1 O MgBr ether 2 H3O 1 O MgBr ether O H 2 H3OPredict the major product(s) of each of the following reactions_ A) Br2 light B) Mglether 2H OH PCC CH2Cl2
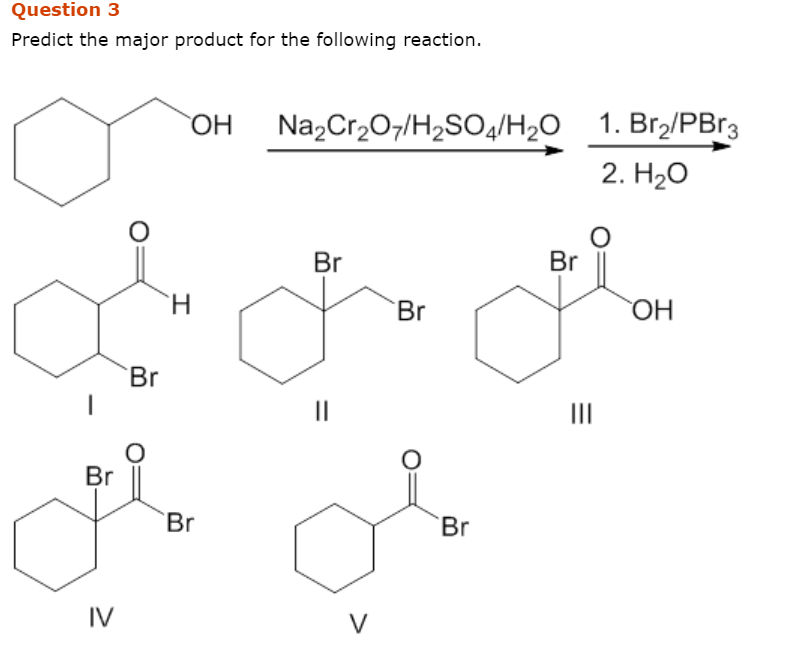



Solved Predict The Major Product For The Following Reaction Chegg Com




Oxidations Chromium Based Jones Pcc Pdc Collins Reagent Chromyl Chloride Youtube




Pyridinium Chlorochromate Pcc Oxidation Of Alcohols
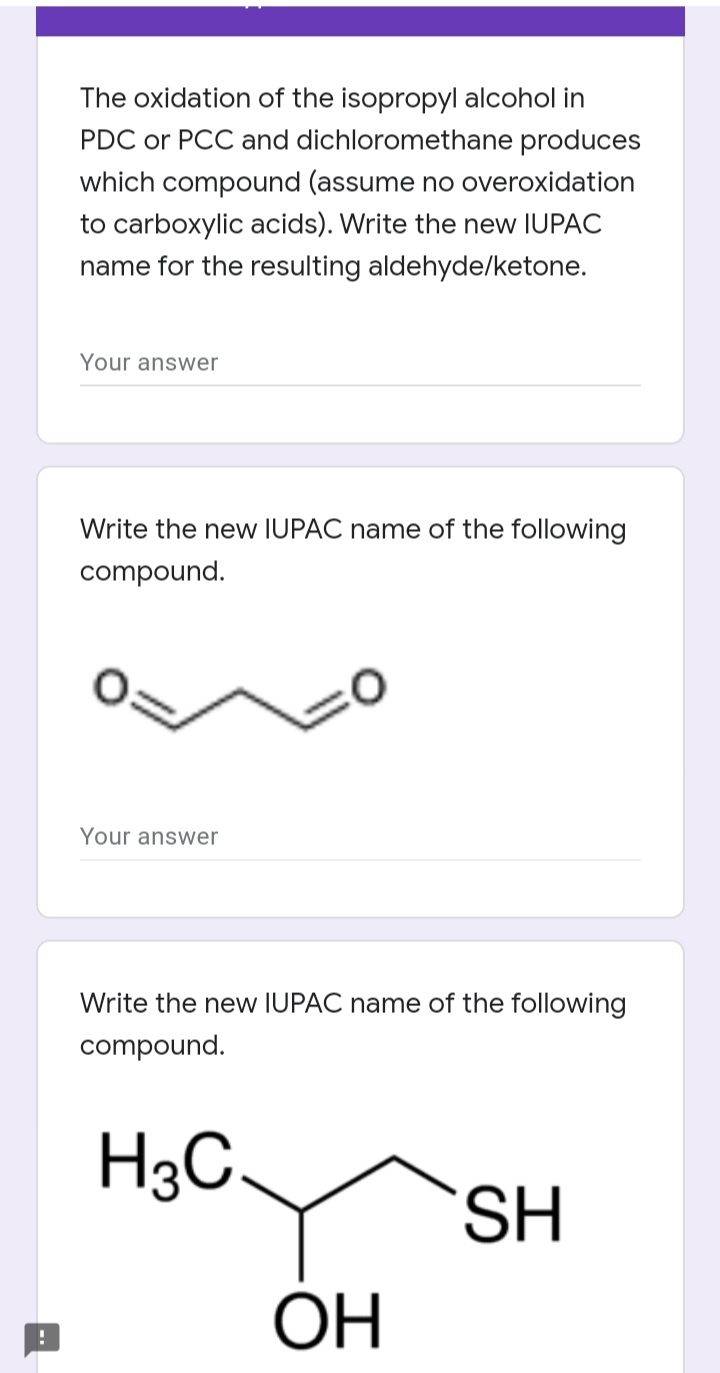



Answered The Oxidation Of The Isopropyl Alcohol Bartleby




Oh H H H Pcc Ch2cl2 O Fe2 H Na2cr2o7 H2so Itprospt
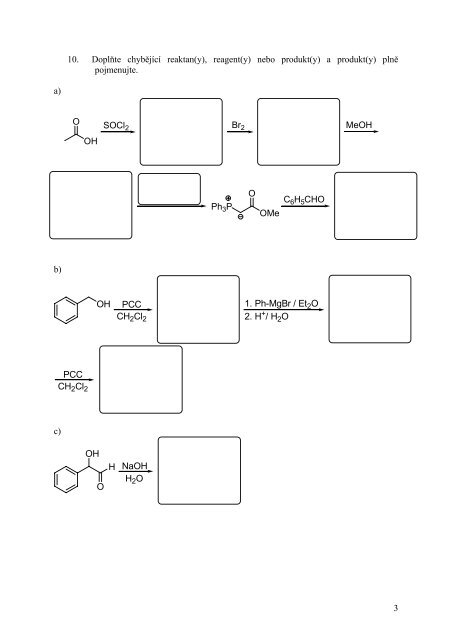



6 Urcete Produkt Y Na



Canvas Wisc Edu




Ch17 C O Prep By Oxidation Of Roh



Solved Question 14 6 Pts Oh Pcc Ch2cl2 Course Hero
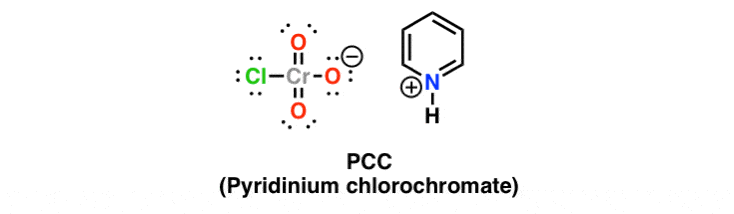



Reagent Friday Pcc Pyridinium Chlorochromate




Nh2 Pcc Ch2cl2 Nabh Cn Oh 8 Br 1 3 Equiv 12 4 Equiv Naoh 1 Homeworklib




Give The Product Expected When The Followi Clutch Prep



Solved 1 Using The Following Alcohol Has Your Only Source Of Carbons Propose A Stepwise Synthesis Of The Target Molecule 2 Propose A Stepwise S Course Hero
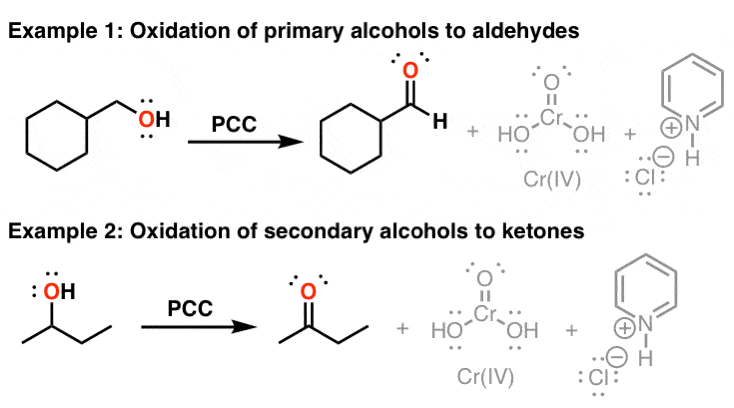



Reagent Friday Pcc Pyridinium Chlorochromate



1
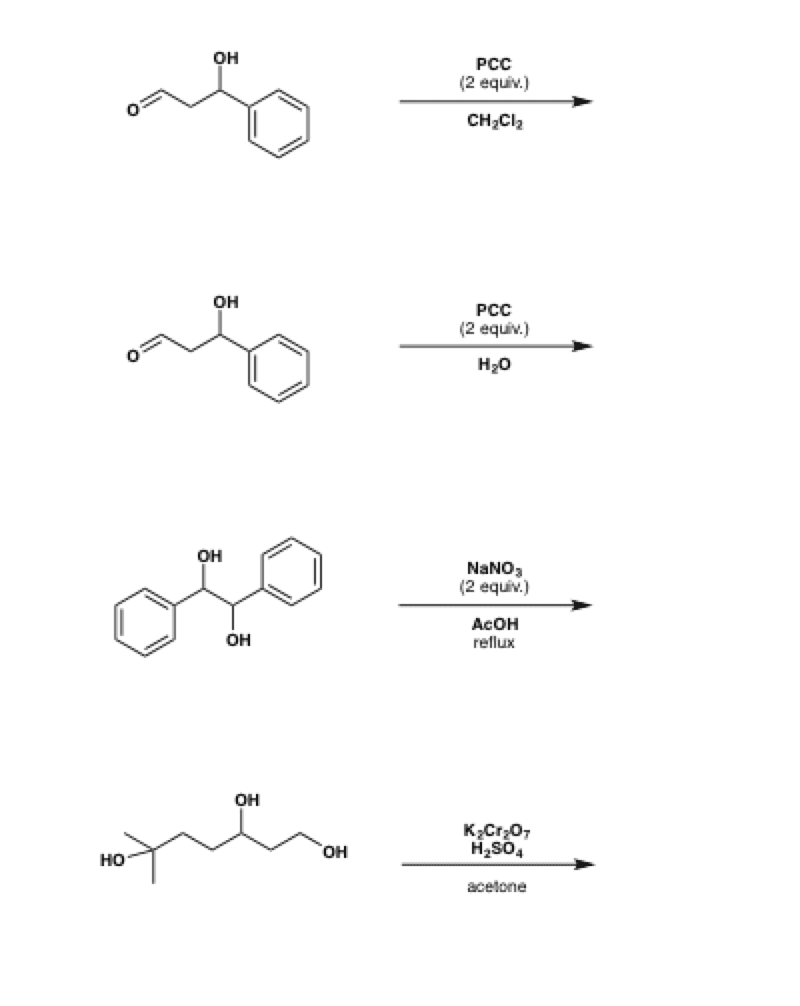



Oneclass Ho Oh Oh Oh Oh Pcc 2 Equiv Ch2cl2 Pcc 2 Equiv H Nano3 2 Equiv Acoh Reflux K2cr 7



Solved Predict The Product For The Following Reaction Oh Chegg Com




Q 3 Single On What Is The Final Product Of The Reaction 1 Bhz Thf Ch3




Organic Chemistry 2 Chapter 19 1 D Quiz 2 2 Flashcards Quizlet



Illustrated Glossary Of Organic Chemistry Pyridinium Chlorochromate Pcc



1



Alcohol Reactions Conversions To Esters Acetate Lab Reactions With Hydrogen Halides Acid Catalyzed Hydrations Ppt Download



Pcc Reagent Definition Preparation Reaction Mechanism




Reagents A I Pcc Ch2cl2 72 Ii Meoch2pph3 59 B I Download Scientific Diagram




Hello Again For Problem 2 I Had To Find Functional Groups When Reacting With Pcc Ch3cl2 For Problem 3 I Had To Name The Alcohols Just Wanna Make Sure My Work Looks




Pyridinium Chlorochromate Wikipedia
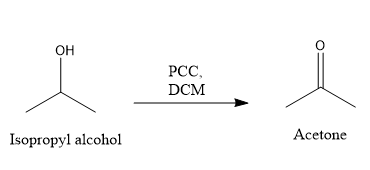



Answered The Oxidation Of The Isopropyl Alcohol Bartleby




Why Can T We Oxidise Allylic Alcohol To Aldehyde Using Pyridinium Chlorochromate Chemistry Stack Exchange
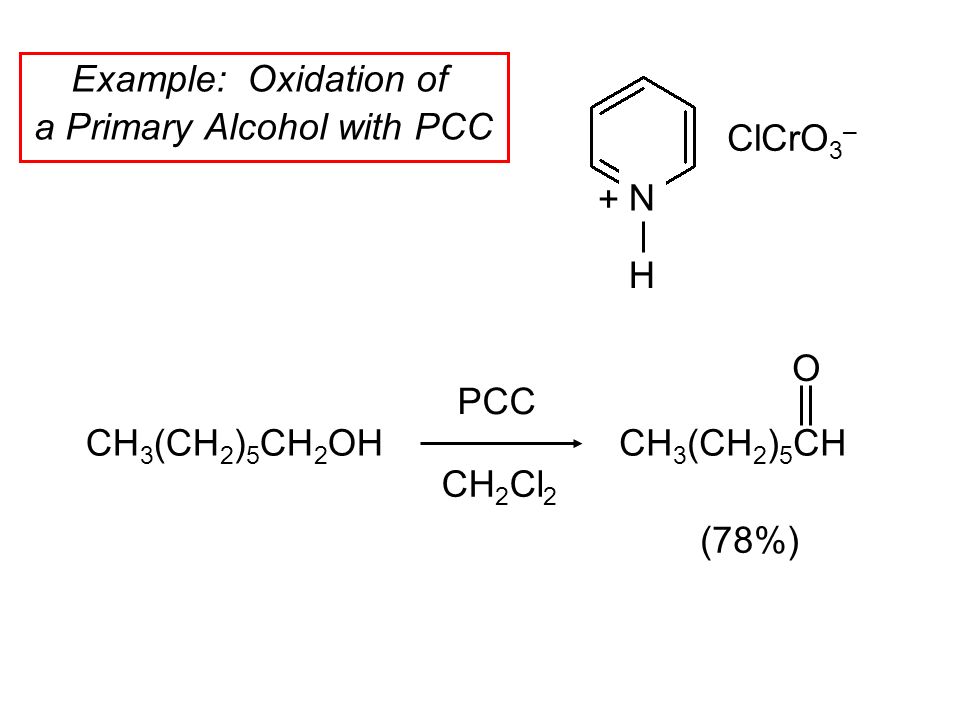



15 6 Reactions Of Alcohols A Review And A Preview Copyright C The Mcgraw Hill Companies Inc Permission Required For Reproduction Or Display Ppt Download



Organic Chemistry On Line




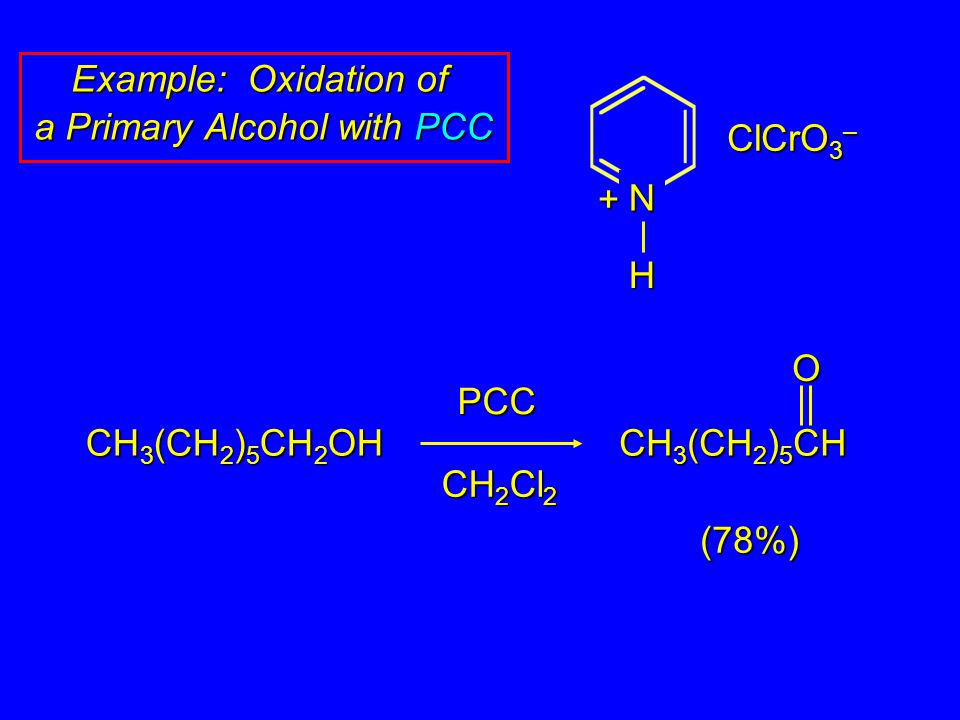
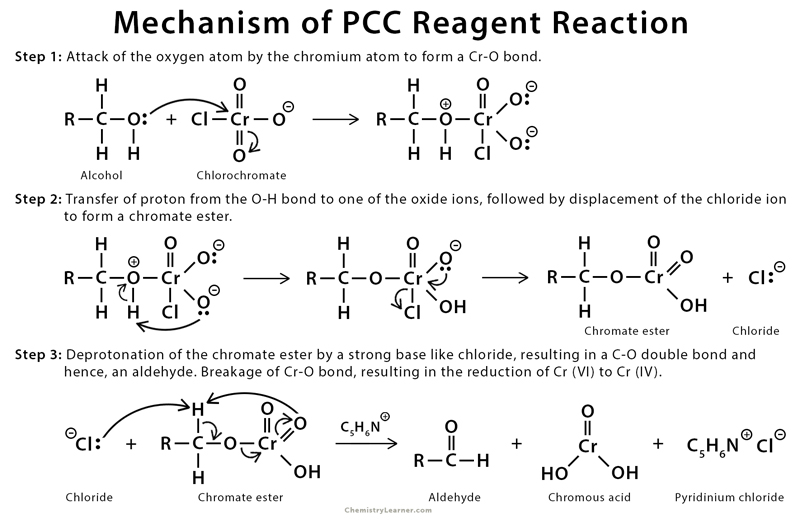
Pcc Oxidation Mechanism Chemistry Steps




Question 14 Predict The Product For The Following Reaction On Pcc Excess Sn2on Ch2cl2 Homeworklib
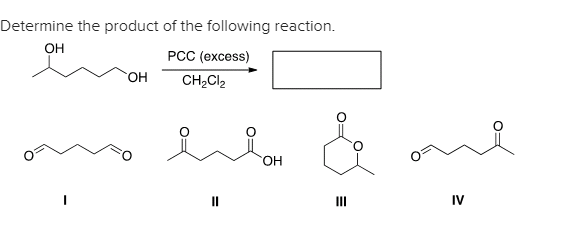



Solved Determine The Product Of The Following Reaction Oh Chegg Com




Determine The Product Of The Following Reaction Pcc Excess Oh Ch2cl2 Oh Oh Oni On Homeworklib




4 H2o 5 Pcc Ch2cl2 Hno3 A Ca Oh 2 Organic Chemistry




What Is The Product When But 2 En 1 Ol Gets Oxidised By Pcc Organic Chemistry Doubts Goiit Com




Using Pcc Organic Chemistry



Solved Oh Led Nochan In For The Na2cr7 H2so4 Oh Pcc Ch2ch2 Provide The Detailed Mechanism For The Reaction Of Benzyl Chloride W Oh Ide Follow Course Hero
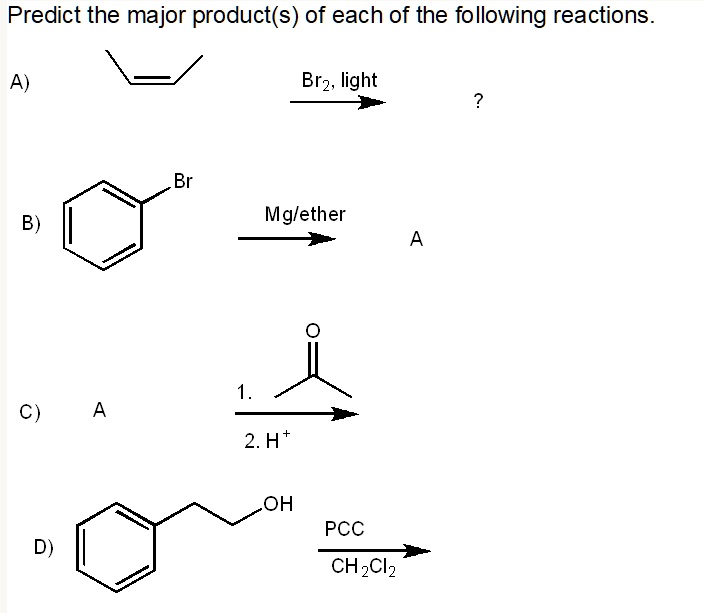



Solved Predict The Major Product S Of Each Of The Following Reactions A Br2 Light B Mglether 2 H Oh Pcc Ch2cl2
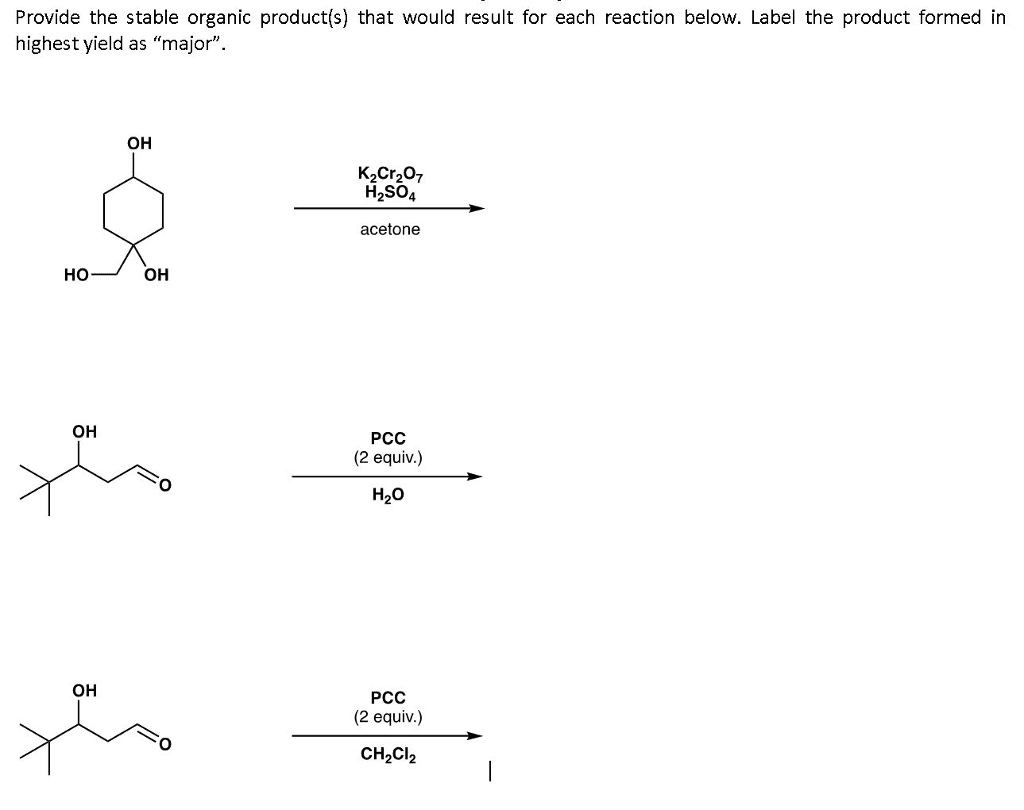



Oneclass Provide The Stable Organic Product S That Would Result For Each Reaction Below Label The



Illustrated Glossary Of Organic Chemistry Pyridinium Chlorochromate Pcc
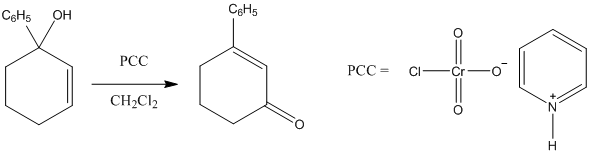



Solved The Tertiary Alcohol Below Was Reacted With Pcc In Ch2cl2 Chegg Com
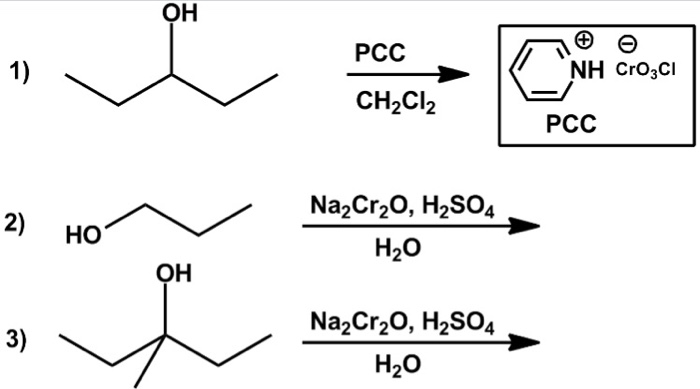



Solved Oh Pcc 1 Nh Crojci Ch2cl2 Pcc Na2cr H2so4 H 2 Chegg Com




Select More Than One Steps Oh Pcc Nabha Ch3oh 1 Mgbr Ch2cl2 Etoh H 2 H Homeworklib
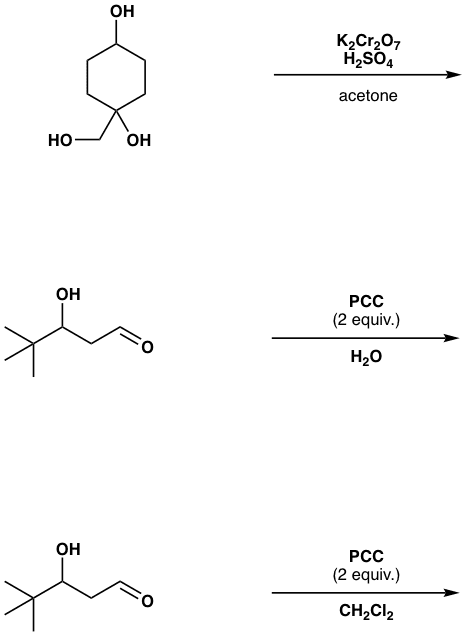



Oneclass Oh H2so4 Acetone Ho Oh Oh Pcc 2 Equiv H Oh Pcc 2 Equiv Ch2cl2




Solved Oh Nah Dmf 1 Pcc Ch2cl2 2 Nah Thf O O Oet Eto Naio4 Chegg Com




4 Pts 18 Aside Pcc Is Made In Situ From Organic Chemistry




Diol Oxidation Example 1 Youtube




Reagents A I Pcc Ch2cl2 72 Ii Meoch2pph3 59 B I Download Scientific Diagram
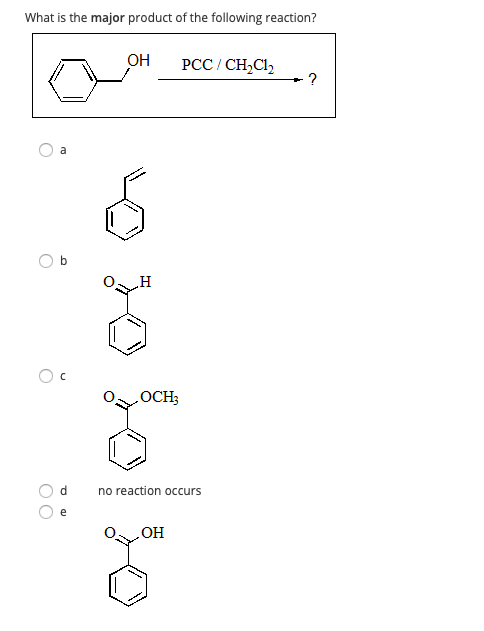



Solved What Is The Major Product Of The Following Reaction Chegg Com



Solved 10 What Would Be The Major Product Of The Following Reaction Sequence Oh Pcc I Lda Ch C12 Ii Chi I Ii Iii Iv 4 A B 1 H Ii




Pyridinium Chlorochromate Pcc
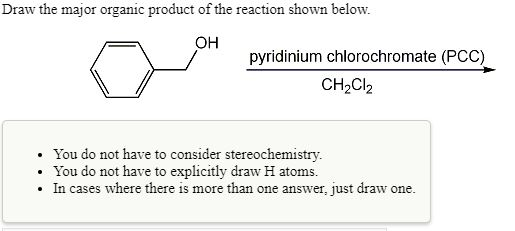



Solved Draw The Major Organic Product Of The Reaction Shown Chegg Com




Chapter 3 Ketones And Aldehydes Flashcards Quizlet
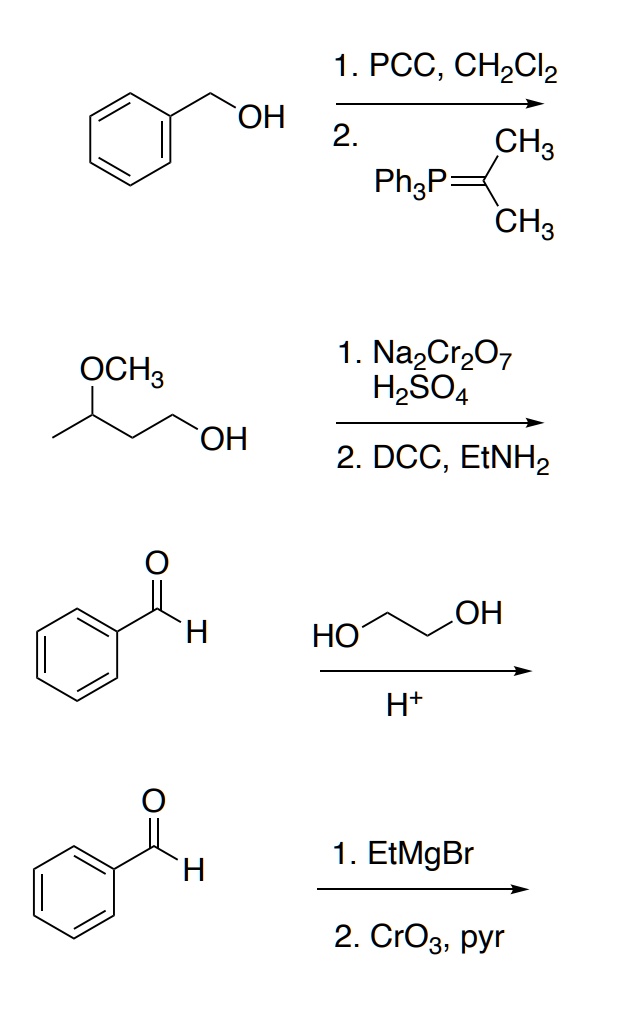



Solved 1 Pcc Chzclz Oh 2 Ch3 Phzp Ch3 1 Nazcr7 H2so4 Oh 2 Dcc Etnhz Oh Ho H 1 Etmgbr 2 Cro3 Pyr Ochs
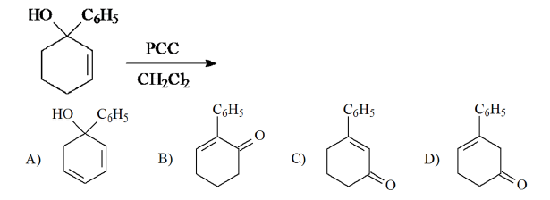



Solved The Tertiary Alcohol Below Was Reacted With Pcc In Ch2cl2 Chegg Com




Pcc Oxidation Mechanism Chemistry Steps
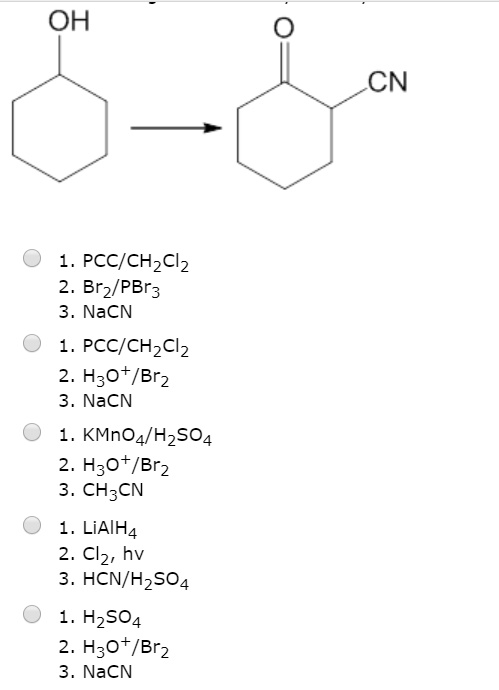



Solved Oh Cn Pccichzclz 2 Brz Pbr3 Njen A Pcc Chzclz 0fh Br2 3 Nacn Kmno4 Hzso4 H3o Brz 3 Ch3cn Liaih4 2 Clz Hv 3 Hcn H2so4 Hzso4 H3o Br2 Njen A



1




Orgo 2 Chapter 11 Flashcards Quizlet
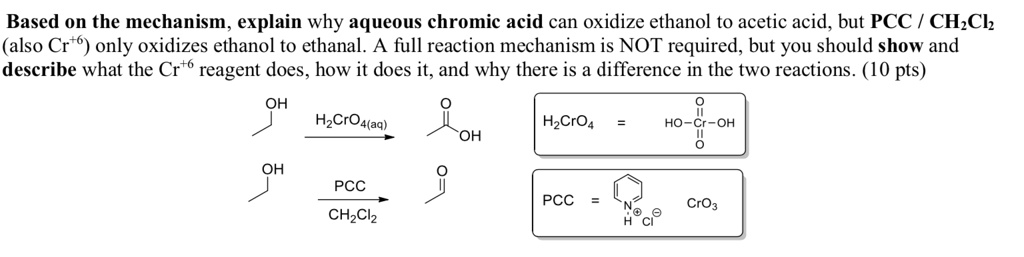



Solved Based On The Mechanism Explain Why Aqueous Chromic Acid Can Oxidize Ethanol To Acetic Acid But Pcc Ch Ch Also Cr 6 Only Oxidizes Ethanol To Ethanal A Full Reaction Mechanism Is




Solved Predict The Product Draw The Major Organic Product For Each Of The Transtutors




Give The Product Expected When The Followi Clutch Prep




Pyridinium Chlorochromate Pcc Oxidation Of Alcohols
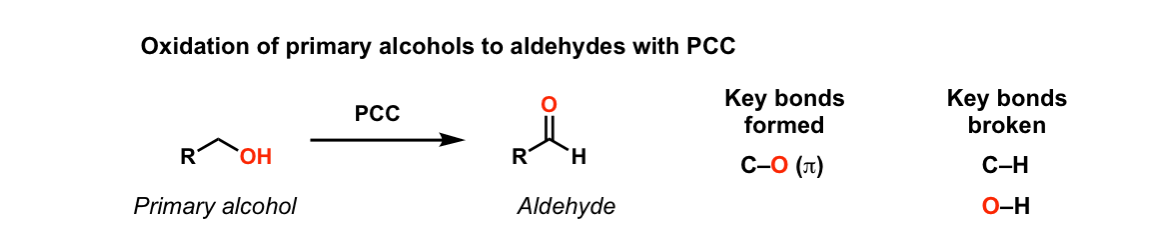



Oxidation Of Primary Alcohols To Aldehydes Using Pcc Master Organic Chemistry



1




Scheme 1 Semisynthesis Of 1 From Vibralactone Reagents And Download Scientific Diagram




Pyridinium Chlorochromate An Overview Sciencedirect Topics
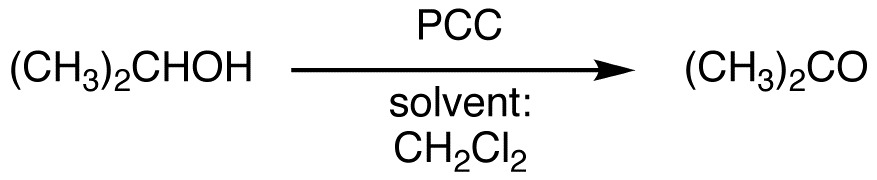



Pyridinium Chlorochromate Chemistry Libretexts




Using Pcc Organic Chemistry
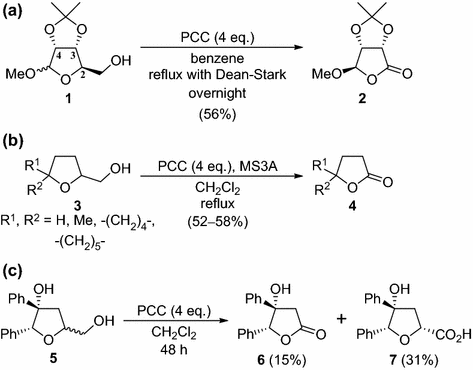



Practical And Environmentally Friendly Transformation Of Tetrahydrofuran 2 Methanols To G Lactones Via Oxidative Cleavage Springerlink
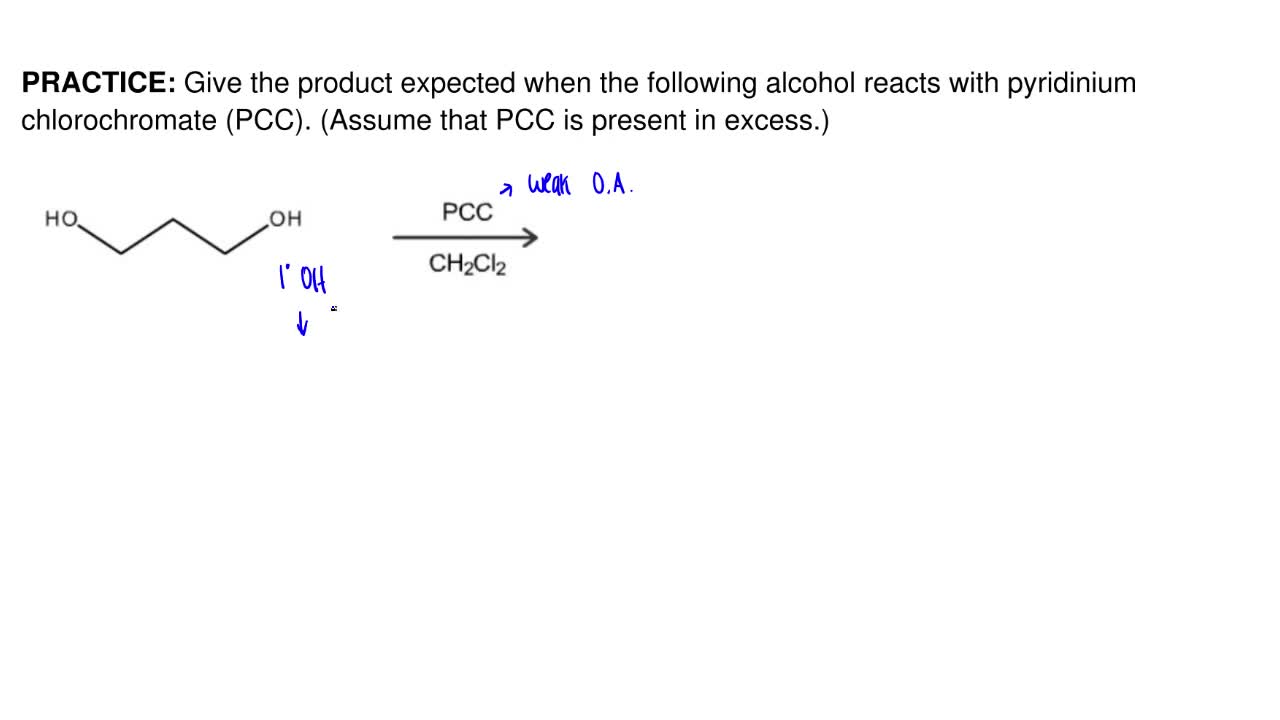



Give The Product Expected When The Followi Clutch Prep




Pyridinium Chlorochromate Pcc Oxidation Of Alcohols
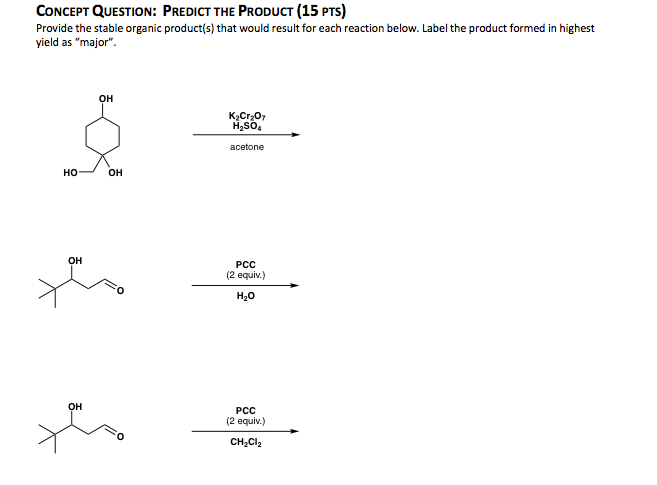



Oneclass Oh H2so4 Acetone Ho Oh Oh Pcc 2 Equiv H Oh Pcc 2 Equiv Ch2cl2




Pyridinium Chlorochromate An Overview Sciencedirect Topics



Solved Determine The Product Of The Following Reaction Course Hero




Ch 21 Test Bank Flashcards Quizlet
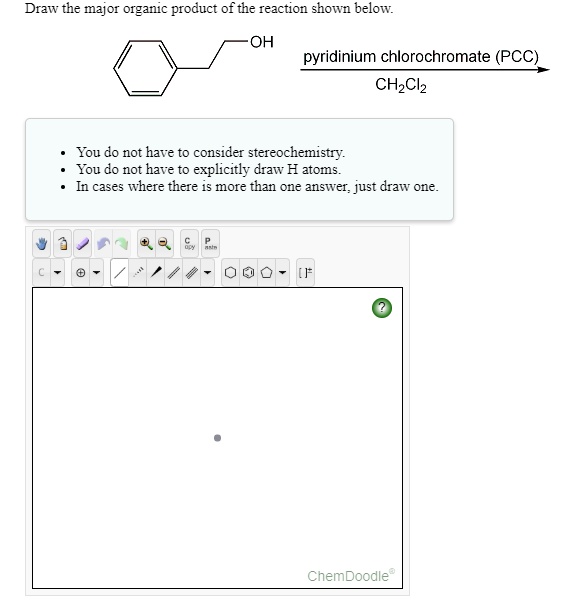



Solved Draw The Major Organic Product Of The Reaction Shown Below Oh Pyridinium Chlorochromate Pcc Chzclz You Do Not Have To Consider Stereochemistry You Do Not Have To Explicitly Draw H Atoms In



0 件のコメント:
コメントを投稿